















Amicapsil’s effect on pressure injuries in SCI
Read more
Read more
Both studies find 100% closure rates
for acute and chronic pressure injuries
are on NHS Supply Chain
quicker than antibiotics and antiseptics.
Effective on resistant infections and does not contribute to AMR.
Effective on resistant infections and does not contribute to AMR.
Effective on resistant infections and does not contribute to AMR.
Review article discussing
the wound microbiome and Amicapsil
Review article discussing
the wound microbiome and Amicapsil
Review article discussing
the wound microbiome and Amicapsil
acting via the skin and wound microbiome without the use of antimicrobials.
acting via the skin and wound microbiome without the use of antimicrobials.
acting via the skin and wound microbiome without the use of antimicrobials.
Effective wound treatment in immunocompetent and immunocompromised persons.
Effective wound treatment in immunocompetent and immunocompromised persons.
Effective on antibiotic and antiseptic resistant wound infections.
Effective in immunocompetent and immunocompromised persons.
The Microbiome Supporting Soap is ideal for sensitive skin, including the skin of babies and toddlers.
The Microbiome Supporting Soap is ideal for sensitive skin, including the skin of babies and toddlers.
The Microbiome Supporting Soap is ideal for sensitive skin, including the skin of babies and toddlers.
“From 4 weeks of absolute hell – to being pain-free, getting a full night’s sleep and back to normal at work.”
“From 4 weeks of absolute hell, in pain 24 hours a day, struggling at work – to being pain-free, getting a full night’s sleep and back to normal at work.”
“From 4 weeks of absolute hell, in pain 24 hours a day, struggling at work – to being pain-free, getting a full night’s sleep and back to normal at work.”
NH ( patient)
NH ( patient)
NH ( patient)
Effective in treating severe infections, including infections not responding to antibiotics and antiseptics.
Effective in treating severe infections, including infections not responding to antibiotics and antiseptics.
Effective in treating severe infections, including infections not responding to antibiotics and antiseptics.
60% quicker removal of infection than antibiotics and antisepics.
60% quicker removal of infection than antibiotics and antisepics.
60% quicker removal of infection than antibiotics and antisepics.
No additives or chemicals
No additives or chemicals
No additives or chemicals
Infection causes pain and by removing the infection, Amicapsil and SertaSil reduce the pain.
Infection causes pain and by removing the infection, Amicapsil and SertaSil reduce the pain.
Infection causes pain and by removing the infection, Amicapsil and SertaSil reduce the pain.
Effective treatment of wounds and pressure ulcers in SCI-persons and in controlling soft tissue infection caused by osteomyelitis.
Effective treatment of wounds and pressure ulcers in SCI-persons and in controlling soft tissue infection caused by osteomyelitis.
Effective treatment of wounds and pressure ulcers in SCI-persons and in controlling soft tissue infection caused by osteomyelitis.
climate change
VH (patient)
VH (patient)
VH (patient)
it is biologically recyclable and does not require the use of chemicals, plastics or antimicrobials.
MPPT - micropore particle technology
MPPT – micropore particle technology is an emerging novel type of treatment for wounds and dermatological skin lesions. It acts via the wound microbiome-immune axis to support the immune system, thereby enabling the body’s immune system to fight wound infection and regenerate new tissue to close the wound.
MPPT uses the micro-evaporation of moisture from the skin and wound surface to transport away microbial toxins and enzymes and to disrupt the structure of biofilm. Disrupting these microbial defence systems, returns control to the immune system such that it can remove the wound infection and regenerate lost tissue. MPPT does not have any antimicrobial effects and nothing is released into the wound.
Studies in humans and animals have shown that MPPT removes wound infections 60% quicker than antibiotics and antiseptics and that these effects lead to stable wound closure. MPPT is effective in immunocompetent and in immunocompromised individuals, including in conditions such as diabetes, spinal cord injury and multiple sclerosis.
MPPT has been found safe to use and has not been associated with allergy or wound irritation.

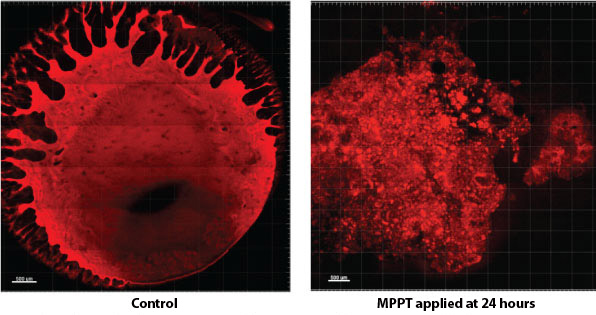
MPPT disrupts mature biofilm
MPPT is a Class II medical device and is CE-marked. It is approved as a treatment for wounds, i.e. with a therapeutic claim similar to pharmaceuticals. It is the only wound product to achieve this.
MPPT is available over-the-counter, i.e. a prescription is not needed, and is available for private use. The bottles are not single use. MPPT is temperature sensitive and needs to be stored on refrigeration.
MPPT is available as Amicapsil and Aprobaxil for human use. SertaSil and Adinasil are for veterinary use.
MPPT only involves natural ingredients that are readily biologically recyclable and only water and cotton are used in the process. All packaging is recyclable.
No contribution to climate change or environmental damage.
Independent review of MPPT (Amicapsil)

Skin Microbiota: A Novel Target for Promoting Burn Wound Healing
Wound Repair and Regeneration publishes extensive international coverage of cellular and molecular biology, connective tissue, and biological mediator studies in the field of tissue repair and regeneration. It is the official journal of The Wound Healing Society (USA), The European Tissue Repair Society, The Japanese Society for Wound Healing, and Wounds Australia.
Quotes from publication (section 7.1)
“Amicapsil removes infections 60% faster than conventional treatments such as antibiotics and antiseptics in a spectrum of wounds, including acute wounds, venous leg ulcers, diabetic foot ulcers, and burns [145]. Studies have confirmed the efficacy of this method across various wound types, resulting in complete and stable wound closure.”
“These findings suggest that wound management strategies targeting the microbiome–immune axis may offer superior efficacy compared to conventional antimicrobial approaches. This approach, which interacts with the microbiome without eradicating microorganisms or cells, offers a promising strategy to effectively address wound infections while supporting regenerative processes.”
Antimicrobials ineffective
FDA (2016): Classification of Wound Dressings Combined with Drugs
“The available evidence does not appear to demonstrate improved clinical outcomes from the use of antimicrobial dressings over non-antimicrobial dressings for the prevention or treatment of local wound infections or to improve wound healing.”
FDA (2022): Advancing product development for non-healing chronic wounds.
“Despite the public health burden, innovative products aimed at the treatment of non-healing chronic wounds are lacking.”
“innovative product development is essential to addressing the unmet medical need of non-healing chronic wounds”.
Benefits of MPPT
- 60% quicker removal of wound infection and 50% quicker onset of tissue regeneration than antibiotics and antiseptics.
- Effective across wound types.
- Effective against antimicrobial resistant wound infections.
- Effective in persons with compromised immune function.
- Reduction in infection reduces wound pain.
- Studies in dehished surgical wounds and pressure ulcers shown 100% healing rates.
- Able to control soft tissue infection caused by underlying osteomyelitis.
- Allows surgery in non-infected tissue.
- Bed rest not required.
- No wound irritation or allergy observed.
- Components non-toxic if ingested.
- Components biologically readily recyclable.
Value of MPPT
Reduced costs-of-care in community:
- Acute pressure ulcers: 80% savings
- Suitable for self-care and telemedicine
- Improved safety and quality of care
- Reduced recurrence of wounds
Improved hospital performance:
- Reduced patient mortality
- Reduced length of stay
- Improved safety and quality of care
- Reduced admission rates
Implementation of MPPT:
- Easy to use.
- Rapid healing supports compliance.
- No allergy or irritation to reduce compliance.
Overview of MPPT publications
| Source | Study | Design | Wound Type | N of wounds | Comparator | Primary outcome | Secondary outcome | Both Achieved | Safety issues | Comments |
|---|---|---|---|---|---|---|---|---|---|---|
| Bilyayeva et al. (2014) | Preclinical | RCT | Preclinical model | 45 | Gentamicin, untreated | Infection removal | Closure | Yes | No | MPPT infection free 60% quicker and reached closure 26% quicker vs. gentamicin. |
| Bilyayeva et al. (2017) | Hospital inpatients | RCT | Acute, SSI, DFU, VLU, burns | 266 | Gentamicin, untreated | Infection removal | Hospital discharge | Yes | No | MPPT infection-free 60%, onset of granulation 50%; and discharge 31% quicker vs. gentamicin. Acute wounds reached substantially quicker full closure compared to gentamicin and iodine. |
| Ryan (2017) | Hospital inpatients* | RWE | SSI | 10 | NPWT | Infection removal | Hospital discharge | Yes | No | Tissue granulation 81% quicker than historic control. All wounds proceeded towards closure. |
| Sams-Dodd et al. (2018) | Community (4 patients, 1 veterinary) | Case | PI, DFU, VLU, trauma | 5 | Standard care | Infection removal | Size reduction or closure | Yes | No | Wound infection removed and healing seen in all cases. Two wounds could be followed to closure, two patients died of non-wound related conditions, and one could not be followed. |
| O'Sullivan et al. (2020) | Rehab-ward patient* | Case | DF | 1 | Aquacel & NPWT | Infection removal | Rehab | Yes | No | Removal of soft tissue infection, large reduction in wound size. Comparators were ineffective. |
| Sams-Dodd et al. (2020) | Community | Case | DF | 1 | Manuka honey, PHMB | Infection removal | Surgery | Yes | No | Removal of soft tissue infection, allowing surgery. Large reduction in wound size. Comparators were ineffective. |
| Sams-Dodd et al. (2024) | Community using telemedicine | RWE | PI/DF | 44 | Standard care | Wound closure and DF control | Costs and resource savings | Yes | No | MPPT achieved 100% closure of acute and chronic wounds, including wounds that had failed to close with standard care. Control of soft tissue infection from DF. Substantial cost and resource savings. |
| Smith and Ridler (2024) | Community* | PRO | PI/DF | 49 | Standard care | Wound closure and DF control | User saisfaction | Yes | No | MPPT achieved 100% closure in acute and chronic wounds. All chronic wounds had prior been unsuccessfully treated with standard care. Control of soft tissue infection from DF. High patient satisfaction with the use of MPPT. |
| Sams-Dodd et al. (2025) | Climate change is assumed to contribute to AMR, but an analysis finds that the reverse is the case, i.e. that AMR strongly contributes to climate change. A single course of antibiotics is estimated to cause the release of 9.84 tonnes of CO2 – the equivalent of a standard car driving around the Earth 1.47 times. Unlike standard care approaches to infected wound, which are known to be ineffective, MPPT involves no antimicrobials and is non-toxic. Therefore, it does not contribute to climate change. | |||||||||
| Sams-Dodd et al. (2026), submitted | Veterinary use | RWE | Necrotizing fasciitis, amputation, trauma, moist dermatitis | 21 | Standard care | Wound closure | High quality of closure and scarring | Yes | No | MPPT achieved 100% closure of acute and chronic wounds. Stable closure was achieved in cases with acute osteomyelitis. All chronic wounds had been unsuccessfully treated with standard care as well as in several cases amputated, including repeat amputation. |
*: independent study. Study designs: PRO: patient-reported outcome study; RCT: Randomised Controlled Trial; RWE: Real-world-evidence study. Wound types: DF: draining fistula; DFU: diabetic foot ulcer; PI: Pressure injury; SSI: surgical site infection; VLU: venous leg ulcer.
Overview of general publications
Articles related to the research performed by Willingsford in general journals or websites.
| Topic | Description | Link |
|---|---|---|
| AMR and Environment | Research show that the use of antimicrobials activate bacterial defence systems, typically making an infection in a surface worse. | Environmental Stewardship - The Role of Antimicrobials in Climate Change |
| Space | The microbial environment aboard a spacecraft is essential to allow deep-space exploration. | https://www.innovaspace.org/blog/populate-the-space-station-with-microbes-to-stay-healthy |
Wounds, ulcers and burns
Amicapsil for humans
Trauma and surgical wounds, pressure ulcers, venous leg ulcers, diabetic foot ulcers, burns, etc.
SertaSil for animals
Trauma and surgical wounds, abscesses, amputations, and burns in dogs, horses and other animals.
Dermatology
Aprobaxil for humans
Acne, pyoderma gangrenosum, hidradenitis suppurativa, atopic dermatitis, zits etc
Adinasil for animals
Eczema, dermatitis, lick granuloma, insect bites, autoimmune skin conditions, etc.
Medical and scientific conferences









Publications and Abstracts
- Sams-Dodd, J. and Sams-Dodd, F. (2026) Environmental Stewardship – The Role of Antimicrobials in Climate Change. Contagion Spring 2026 Digital Edition, page 36-40. Visit ContagionLive
- Sams-Dodd, J. and Sams-Dodd, F. (2025) Populate the space station with microbes to stay healthy. InnovaSpace Blog.
- Sams-Dodd, J. and Sams-Dodd, F. (2025) The contribution of antimicrobials and antimicrobial resistance to climate change and a possible way to reverse it whilst still offering high quality healthcare—a conceptual analysis. Frontiers in Public Health
- Smith, D. and Ridler, M. (2024) Patient-reported outcome survey of user-experiences in the spinal cord injured-community with MPPT for treating wounds and pressure injuries and for controlling soft tissue infection caused by osteomyelitis. Frontiers in Rehabilitation Sciences
- Sams-Dodd, J., Belci, M., Bandi, S., Smith, D., and Sams-Dodd, F. (2024) Stable closure of acute and chronic wounds and pressure ulcers and control of draining fistulas from osteomyelitis in persons with spinal cord injury: non-interventional study of MPPT passive immunotherapy delivered via telemedicine in community care. Frontiers in Medicine
- Smith, D (2023) Addressing the challenges of treating pressure ulcers following spinal cord injury with Amicapsil-SCI micropore particle technology.ISCoS, Edinburgh, 8’th to 11’nd October, abstract #56. See Abstract and Poster
- Sams-Dodd, F. and Sams-Dodd, J. (2023) MPPT for controlling soft tissue infection caused by osteomyelitis. ISCoS, Edinburgh, 8’th to 11’nd October, abstract #98.
- Sams-Dodd, F. and Sams-Dodd, J. (2023) Diagnosing osteomyelitis using wound characteristics and response to MPPT. ISCoS, Edinburgh, 8’th to 11’nd October, abstract #187.
- Sams-Dodd F. and Sams-Dodd, J. (2023) Effective treatment of wound infections using physical interactions with the microbiome – without the use of antimicrobials. Online symposium May 22-23, 2023, University of Gibraltar.
- Sams-Dodd, F. and Sams-Dodd, J. (2023) Evaluating a passive immunotherapy acting via the microbiome for treating injured and infected dog tails: a prospective case-series. BSAVA, Manchester, March 23-25.
- Sams-Dodd, F., Belci, M., Smith, D. and Sams-Dodd, J. (2022) MPPT, delivered by a telemedicine approach, for treating acute and chronic wounds and pressure ulcers in SCI-persons. ISCoS, Vancouver September 15-18, abstract #295.
- Sams-Dodd, F. and Sams-Dodd, J. (2022) Effective Treatment of Burn Infections by Supporting the Immune System and the Microbiome. MHSRS, September 12-16, Orlando, Fl. USA, abstract MHSRS-22-05489.
- Sams-Dodd, F. and Sams-Dodd, J. (2022) Antimicrobial Development to Counter Wound Infections in Military Personnel. MHSRS, September 12-16, Orlando, Fl. USA, abstract MHSRS-22-05487.
- Sams-Dodd, J., Belci, M., Bandi, S., Smith, D. and Sams-Dodd, F. (2021) Micropore particle technology – new treatment for wounds and pressure ulcers in SCI-persons. ANZSCoS, October 27-29, 2021, Session 3A.
- Sams-Dodd, J., Belci, M., Bandi, S., Smith, D. and Sams-Dodd, F. (2021) Micropore particle technology – new treatment for wounds and pressure ulcers in SCI-persons. EPUAP, September 14-16, Abstract FPS3.1
- O’Sullivan O, Hayton L, Findlay-Cooper K, Phillip R. Novel micropore particle technology for spinal cord injury chronic wound healing: a new paradigm? BMJ Mil Health. 2020 Aug 4. Epub ahead of print.
- Sams-Dodd J and Sams-Dodd F. (2020) Micropore Particle Technology Promotes Wound Healing, Whereas Polyhexamethylene Biguanide Causes Tissue Degeneration: A Case Report. Wounds 32(3):E6–E10.
- Sams-Dodd, F. and Sams-Dodd, J. (2019) Micropore particle technology effectively treats infected wounds and pressure injuries in patients with spinal cord injury. ISCoS, Nice Nov. 5-7, 2019.
- Sams-Dodd J and Sams-Dodd F. (2018) Time to Abandon Antimicrobial Approaches in Wound Healing: A Paradigm Shift. Wounds 30(11):345–352.
- Lovgren ML, Wernham A, James M, Martin-Clavijo A. Pyoderma gangrenosum ulcers treated with novel micropore particle technology. Br J Dermatol. 179(Suppl 1):BI22, 152.
- Ryan E (2017) The use of a micropore particle technology in the treatment of acute wounds. Journal of Wound Care 26(7): 404-413.
- Ebony Ryan, Bristol University Hospital: Clinical evaluation of a novel MMP wound care product on wounds to heal by secondary intervention. TVS 2017 – The Conference, Poster no. 1.
- Sams-Dodd, J. and Sams-Dodd, F. (2017) MPP inactivation of bacterial defences: 60% reduction of inflammatory phase and faster healing. TVS 2017 – The Conference, Poster no. 3.
- Bilyayeva O, Neshta VV, Golub A, Sams-Dodd F. (2017) Comparative Clinical Study of the Wound Healing Effects of a Novel Micropore Particle Technology: Effects on Wounds, Venous Leg Ulcers, and Diabetic Foot Ulcers. Wounds Epub 2017 May 25.
- Bilyayeva O, Neshta VV, Golub A, Sams-Dodd F. (2014) Effects of SertaSil on wound healing in the rat. J Wound Care. 23(8):410, 412-4, 415-6.
- Bilyayeva, O.O; Neshta, V.V., Golub, A.A; Sams-Dodd, F. (2011) Promotion of wound cleansing and acceleration of healing by antimicrobial sorption. EWMA May 23-25, 2011. Abstract P42.
