 |
 |
 |
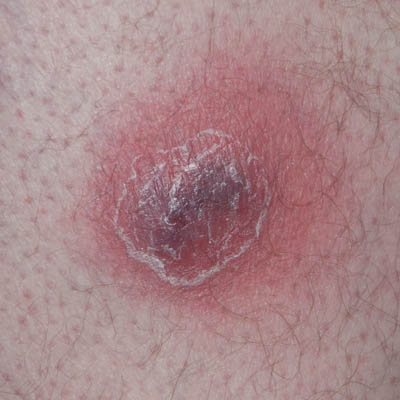
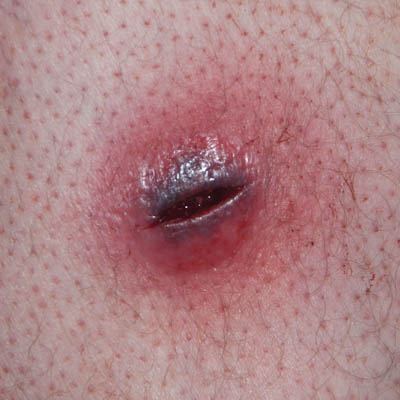
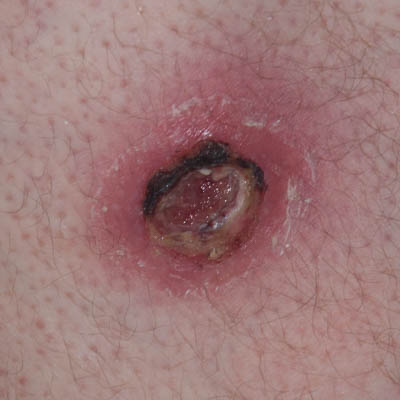
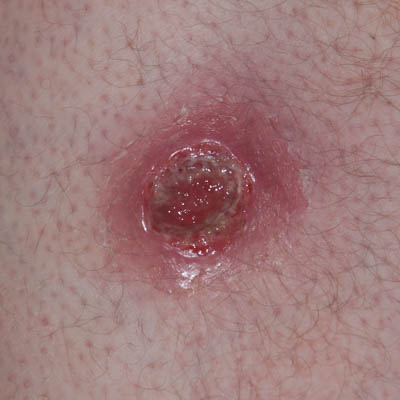
| Day 0 | Day 0 – 1.5 hours after opening | Day 1 |
 |
 |
 |
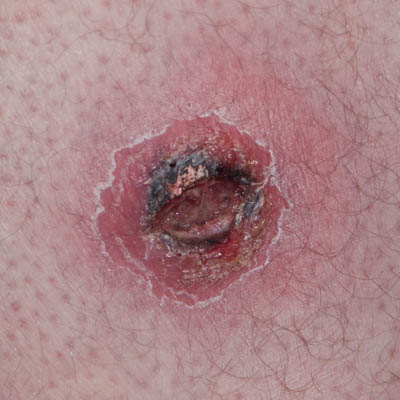
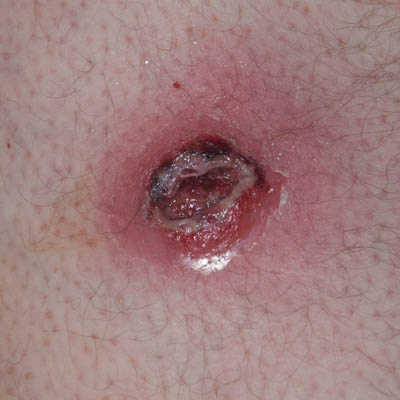
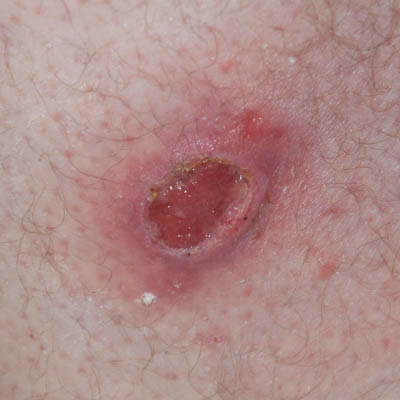
| Day 2 | Day 4 | Day 6 |
 |
 |
 |
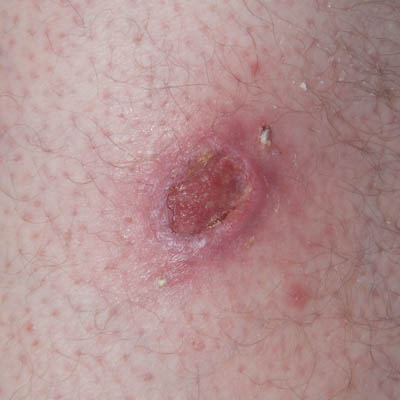
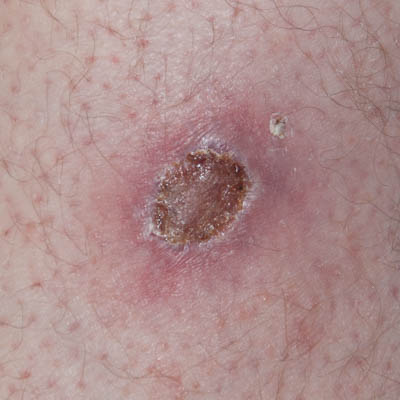
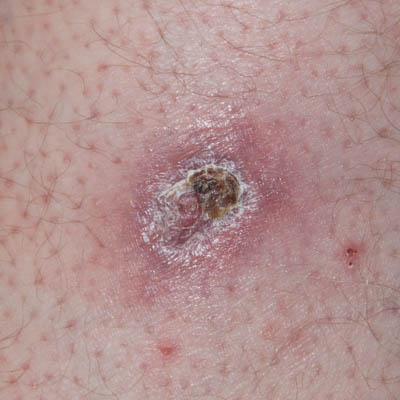
| Day 11 | Day 19 | Day 23 |
 |
 |
 |
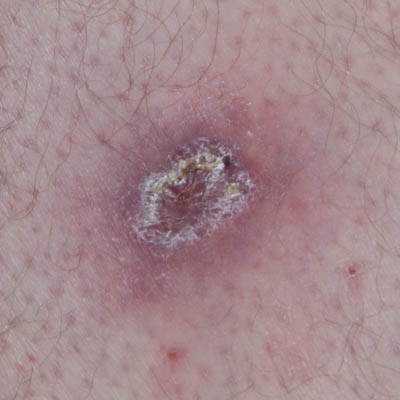
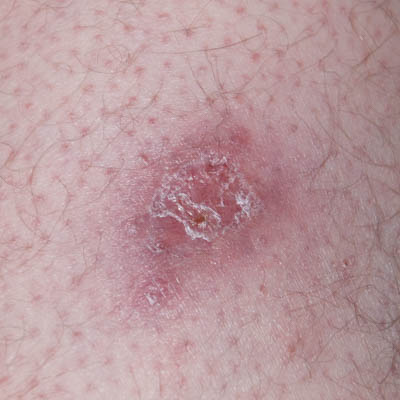
| Day 26 | Day 33 | Day 40 |
 |
 |
 |
| Day 43 | Day 60 | 2 years |
On Day 0, upon surgical opening of the abscess, it was drained of liquefied black blood and necrosis. The abscess was 2 cm deep and the inside of the abscess would consist of soft, non-viable tissue. No type of surgical debridement was performed as the extent of the necrosis induced by the toxins clearly still in the tissue was impossible to determine and would likely have caused extensive bleeding. Indications were, that the toxin-affected area was extensive based on the large inflamed peri-wound area and the rapid enlargement of the abscess but a defined border between viable and non-viable tissue was unclear. The wound was cleaned with isopropyl-alcohol (70%), then tap-water and Amicapsil was applied. As the day progressed, the entire inside of the now opened abscess was rapidly blackening and necrotizing. Late in the day, the wound was filled with liquefied black discharge. It was washed with water and Amicapsil applied.
On Day 1, the patient’s overall state was much improved, the fever had resolved and the peri-wound redness had already reduced. This was taken as an indication that the ongoing expansion of the abscess had ceased.
On Day 1 and 2, due to the reluctance to perform surgical debridement, it was decided to perform very light chemical debridement, i.e. by the use of hydrogen peroxide (3%), applied 2 times 10 seconds followed by thorough washing with tap water. This would be followed by Amicapsil application and covered with a 4-ply lint-free swab (gauze pad).
On the following 2 days (Days 3 and 4) the wound would be washed and Amicapsil applied twice daily. After this, washing and Amicapsil application would be performed once daily.
Over this 5 day period (Days 0 to 5), the distinction between viable and non-viable tissue would gradually become clear. The necrotic tissue would turn black and be removed via autolytic debridement and the peri-wound inflammation would reduce considerably.
On day 6, the wound bed and sides were debrided sufficiently to show fully viable tissue and the wound could be sized up to a 2.5 cm deep area with an opening of 3 x 4 cm.
From Day 1 to25 necrotic slough and waste would every day be present on the secondary dressing (4-ply gauze pad) and be lifted out of the wound with the removal of this. This was taken as an indication that the toxins were still present in the deep tissue and that the potential damage was being kept at bay by the immune system supported by Amicapsil. This assumption was supported by the ability to reduce the peri-wound inflammation while the sides of the wound remained stiff and elevated.
On Day 25, the wound had granulated up fully to level with the wound edges which were no longer elevated and stiff. Furthermore, the inflamed peri-wound area had reduced considerably and together these signs were taken as an indication of the influence of the toxins on the entire area having reduced sufficiently for the healing to progress unaided.
From Day 26-29, washing and Amicapsil application would still be performed to prevent any potential set-back.
Day 30-32 the wound was dressed with a silicone open mesh contact layer dressing. However, this seemed to impede healing and on Day 33 the wound was left uncovered for a few hours. This would dry out the area and facilitate a light natural scab to cover the wound. After a few hours, the wound was left uncovered under the trousers to heal. It was inspected every 1.5 weeks and at no point in time did it need any attention. On Day 60 the wound was closed.
Patient was seen 2 years later for reasons not associated with the wound. The soft tissue had continued to build up beneath the skin all this time and the scar was now leveling with the surface of the surrounding thigh. Also, the scar had continued to reduce in surface area.
The patient had experienced no wound infections or other complications and there had been no soreness of the area since closure. The area had been slightly itchy for the first 6 months after closure on days with very hot and humid weather.













